At Dapsone it is an active ingredient belonging to the group of sulfones. The substance has antimicrobial and anti-inflammatory effects. Dapsone is primarily administered as a tablet and is used to combat rheumatism, acne, blistering skin diseases, inflammatory skin diseases as well as malaria or leprosy.
What is Dapsone?
Dapsone is a drug that has anti-inflammatory and antibacterial effects. The substance belongs to the group of sulfones. Due to its antibacterial effects, it is also counted among the anti-inflammatory drugs. The medicinal substance was first synthesized in Germany in 1908 and patented by I. G. Farben in 1934.
The white (sometimes yellowish white) powder is often used in tablet form and is used to treat rheumatic diseases. It is also often given preventively to combat leprosy, inflammatory skin diseases, malaria, and Pneumocystis pneumonia in patients who are HIV-positive.
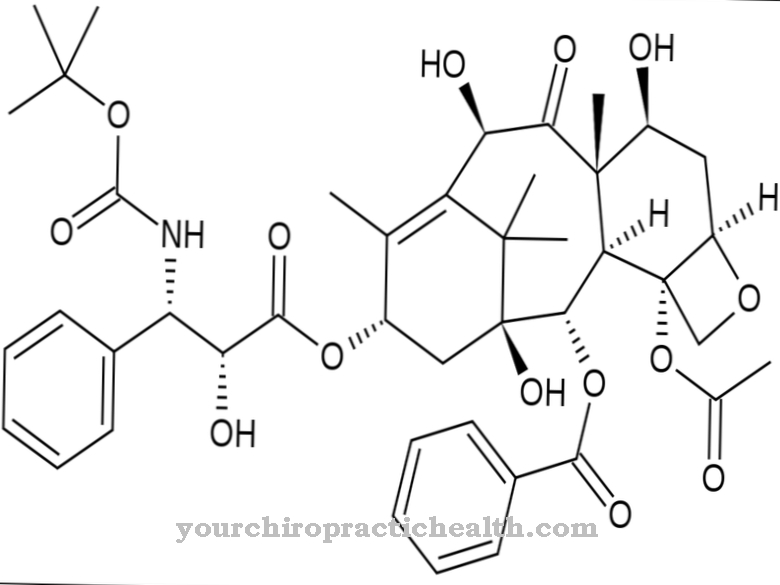
In addition to the name Dapsone, the synonyms are also used Diphenasone, Diaminodiphenyl sulfone, DDS and Dapsonum used to describe the substance. The chemical formula of the drug is C12-H12-N2-O2-S, Mr, which corresponds to a moral mass of 248.3 g / mol.
While in Europe there is only approval in tablet form, in the USA it can also be used as a gel. Here it is used to treat acne.
Pharmacological effect
Dapsone has antibacterial effects against the bacteria Mycobacterium tuberculosis and Mycobacterium leprae (pathogens that cause leprosy) as well as against streptococci. It is also effective against parasites (atiparasitic effect), protozoa and plasmodia. Dapsone also inhibits inflammation.
Dapsone has a direct effect on the folic acid synthesis of bacteria. This creates an antimetabolic inhibition of folic acid synthesis, which ultimately leads to the killing of the bacteria concerned. Since there have been various developments in resistance to the drug since the first administration of Dapsone, the active ingredient is primarily used in combination therapy. Patients therefore usually also take preparations that contain clofazimin or rifampin.
As the listing of Dapsone in the European Regulation (EU) No. 37/2010 shows, the substance may not be used in animals that are processed into food in the European Union. This list is justified by the fact that the substance has high maximum residue levels.
Medical application & use
Due to its wide spectrum of effectiveness, Dapsone has a very broad field of application in human medicine. In the European Union there is only approval for dispensing in tablet form. It is indicated for the treatment of inflammatory skin diseases as well as those that cause blisters. There is also an indication for chronic rheumatic diseases of the joints (rheumatoid arthritis), malaria, rheumatism, leprosy and Pneumocystis pneumonia (a special causative agent of pneumonia in humans).
The medical term of the indication describes that administration for therapeutic purposes is indicated from a medical point of view. To combat Pneumocystis pneumonia, Dapsone is primarily used in patients who are HIV positive. In the treatment of leprosy diseases, Dapsone is always combined with other drugs.
In the United States of America there is also approval for administration as a gel. This is applied locally to the skin to treat acne vulgaris. In addition, other areas of application are recommended in the specialist medical literature, for which, however, there is still no approval, so that no administration has yet taken place in practice.
Risks & side effects
As with most drugs, the administration of preparations containing dapsone can lead to undesirable side effects. Before each initial application, it should therefore be checked whether there is an intolerance or hypersensitivity to dapsone and similar substances. For example, there is a contraindication for hypersensitivity to sulfonamides or for severe liver diseases. This means that from a medical point of view, it is imperative to refrain from using it, as there is a contraindication.
In addition, special caution is required when taking dapsone if there is a hereditary deficiency in glucose-6-phosphate dehydrogenase, anemia, the lips or fingernails discolour bluish, skin rashes or pregnancy occurs. It is imperative to stop breastfeeding while breastfeeding.
There are also interactions with omeprazole (drug that inhibits the formation of gastric acid), trimethoprim (antibacterial drug), rifampicin (tuberculosis drug), pyrimethamine (malaria drug) and ursodeoxycholic acid (preparation for dissolving gallstones).
If these medications are being taken, the attending physician must be informed immediately. During therapy with Dapsone, headaches, stomach upset or nausea can occur frequently (affects 1 to 10 patients in 1000).
In rare cases, bluish discoloration of the skin and anemia (lack of protein in the blood) occur. Rarely means that this was the case in 1 to 10 patients in 10,000. Dapsone syndrome can develop, especially in poorly nourished patients.
Very rarely there has been a depletion of white blood cells (agranulocytosis). See seldom means fewer than 1 patient in 10,000 did. If side effects occur, inform the attending physician immediately. The instructions should be followed urgently.



.jpg)








.jpg)